The outstanding properties of graphene, which have made it one of the most studied materials, can be tailored and enhanced by interaction with other atomic species. In particular, the decoration of graphene with metals is relevant for hydrogen storage applications. Indeed, it is known that graphene cannot store molecular hydrogen at room temperature while it is able to bind atomic hydrogen. Besides, the distance between two stacked graphene layers is too small to allow the hydrogen permeation. Here, metal decoration is useful in several ways: (a) it allows the direct interaction of graphene with molecular hydrogen, via the Kubas interaction, (b) it promotes the catalytic splitting of molecular hydrogen, and (c) it increases the layer spacing between graphene sheets through its intercalation, allowing molecular hydrogen permeation.
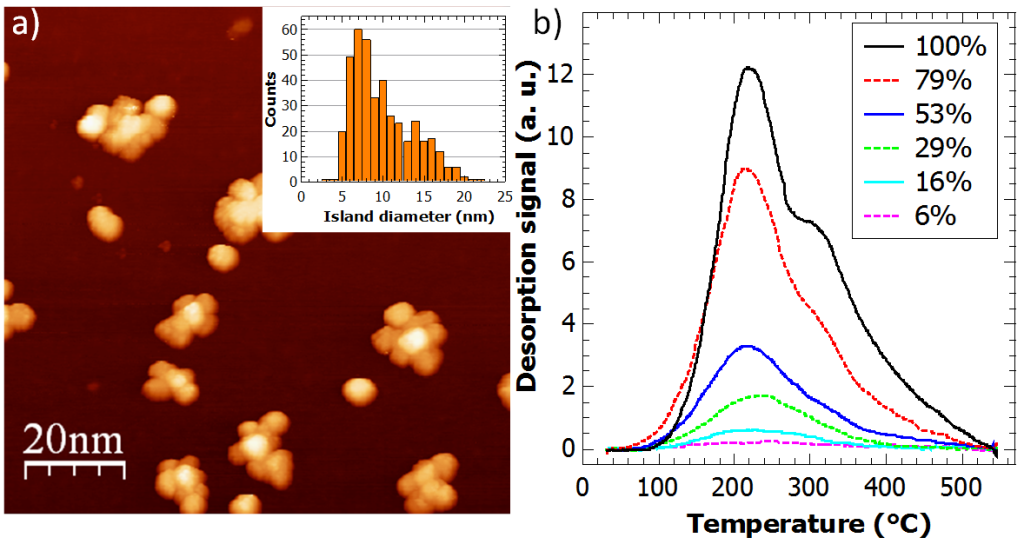
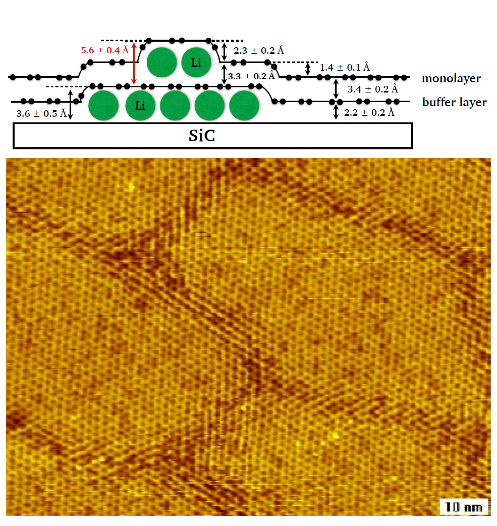
Epitaxial graphene represents an ideal model system to investigate both metal intercalation and its interaction with hydrogen. Our attention is focused on transition metals (Ti, Pt) and alkali metals (Li, Rb). Indeed, transition metals are promising to promote molecular hydrogen storage (see Fig. 1) and the spillover effect via catalytic splitting, while alkali metals are easy to intercalate, at relatively low temperature, providing a graphene layer spacing very close to that ideal for hydrogen permeation (see Fig. 2). For more details, see http://web.nano.cnr.it/heun/research/graphene/.
| People | Letizia Ferbel, Stefan Heun, Stefano Veronesi*, Ylea Vlamidis, Emanuele Pompei |
| Keywords | Hydrogen storage, graphene, metal functionalization |
| Methods, techniques | STM, LEED, TDS, Raman spectroscopy. |
| Collaborations | Chia-Hao Chen (National Synchrotron Radiation Research Center, Hsinchu, Taiwan) and Chung-Lin Wu (Department of Physics, National Cheng Kung University, Tainan, Taiwan) |
| Publications | |
| J.W. Chen, S.H. Hsieh, S.S. Wong, Y.C. Chiu, H.W. Shiu, C.H. Wang, Y.W. Yang, Y.J. Hsu, D. Convertino, C. Coletti, S. Heun, C.H. Chen, C.L. Wu Hydrogen Spillover and Storage on Graphene with Single-Site Ti Catalysts ACS Energy Letters 2022 | |
| L. Ferbel, S. Veronesi, S. Heun: Rb-induced (3×1) and (6×1) reconstructions on Si(111)-(7×7): A LEED and STM study, Surf. Sci. 2022 | |
| S. Veronesi, T. Papa, Y. Murata, S. Heun An atomically flat single-crystalline gold film thermometer to study heat (energy) exchange at the nano-scale App. Surf. Sci. 2020 | |
| Y. Murata, S.Veronesi, D. Whang, S. Heun Morphology of Ti on Monolayer Nanocrystalline Graphene and Its Unespectedly Low Hydrogen Adsorption J. Phys. Chem. C 2019 | |
| L. Basta, S.Veronesi, Y. Murata, Z. Dubois, N. Mishra, F. Fabbri, C. Coletti, S. Heun A Sensitive calorimetric technique to study energy (heat) exchange at the nano-scale Nanoscale 2018 | |
| S. Fiori, Y. Murata, S. Veronesi, A. Rossi, C. Coletti, S. Heun Li-intercalated Graphene on SiC(0001): an STM study Phys. Rev. 2017 | |
| J.-W. Chen, H.-C. Huang, D. Convertino, C. Coletti, L.-Y. Chang, H.-W. Shiu, C.-M. Cheng, M.-F. Lin, S. Heun, F. S.-S. Chien, Y.-C. Chen, C.-H. Chen, and C.-L. Wu, Efficient n-type doping in epitaxial graphene through strong lateral orbital hybridization of Ti adsorbate, Carbon 2016 | |
| K. Takahashi, S. Isobe, K. Omori, T. Mashoff, D. Convertino, V. Miseikis, C. Coletti, V. Tozzini, and S. Heun Revealing the multibonding state between hydrogen and graphene-supported Ti clusters, J. Phys. Chem. C 2016 | |
| T. Mashoff, D. Convertino, V. Miseikis, C. Coletti, V. Piazza, V. Tozzini, F. Beltram, and S. Heun Increasing the active surface of titanium islands on graphene by nitrogen sputtering, Appl. Phys. Lett. 2015 | |
| T. Mashoff, M. Takamura, S. Tanabe, H. Hibino, F. Beltram, and S. Heun Hydrogen storage with titanium-functionalized graphene, Appl. Phys. Lett. (2013) |